|
For example, diethyl ether is the ether with an ethyl group on each side of the oxygen atom. If the two alkyl groups are identical, the ether is called di ether. The most common way is to identify the alkyl groups on either side of the oxygen atom in alphabetical order, then write “ether.” For example, ethyl methyl ether is the ether that has an ethyl group and a methyl group on either side of the oxygen atom. The substituents can be, but do not need to be, the same. An ether is characterized by an oxygen bonded to two alkyl or aryl groups, represented here by and. Nomenclature of Ethers Ethers: The general structure of an ether. Thus, the alpha hydrogens are more acidic than in regular hydrocarbon chains. The oxygen of the ether is more electronegative than the carbons. In organic chemistry, the most common functional groups are carbonyls (\text distances being about 140 pm. 
In materials science, functionalization is employed to achieve desired surface properties functional groups can also be used to covalently link functional molecules to the surfaces of chemical devices. Through routine synthesis methods, any kind of organic compound can be attached to the surface. Functionalization refers to the addition of functional groups to a compound by chemical synthesis. Often, compounds are functionalized with specific groups for a specific chemical reaction. Alkyl chains are often nonreactive, and the direction of site-specific reactions is difficult unsaturated alkyl chains with the presence of functional groups allow for higher reactivity and specificity. Functional Groups and Reactivityįunctional groups play a significant role in directing and controlling organic reactions. 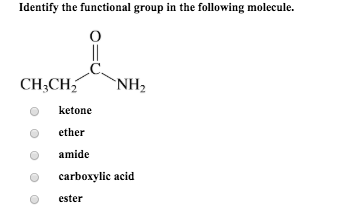
They can be classified as primary, secondary, or tertiary, depending on how many carbon atoms the central carbon is attached to. Classification of alcohols: Alcohols are a common functional group (-OH). 
Similarly, a functional group can be referred to as primary, secondary, or tertiary, depending on if it is attached to one, two, or three carbon atoms. The first carbon atom that attaches to the functional group is referred to as the alpha carbon the second, the beta carbon the third, the gamma carbon, etc. The atoms of a functional group are linked together and to the rest of the compound by covalent bonds. Functional groups also play an important part in organic compound nomenclature combining the names of the functional groups with the names of the parent alkanes provides a way to distinguish compounds. The same functional group will behave in a similar fashion, by undergoing similar reactions, regardless of the compound of which it is a part. In organic chemistry, a functional group is a specific group of atoms or bonds within a compound that is responsible for the characteristic chemical reactions of that compound. 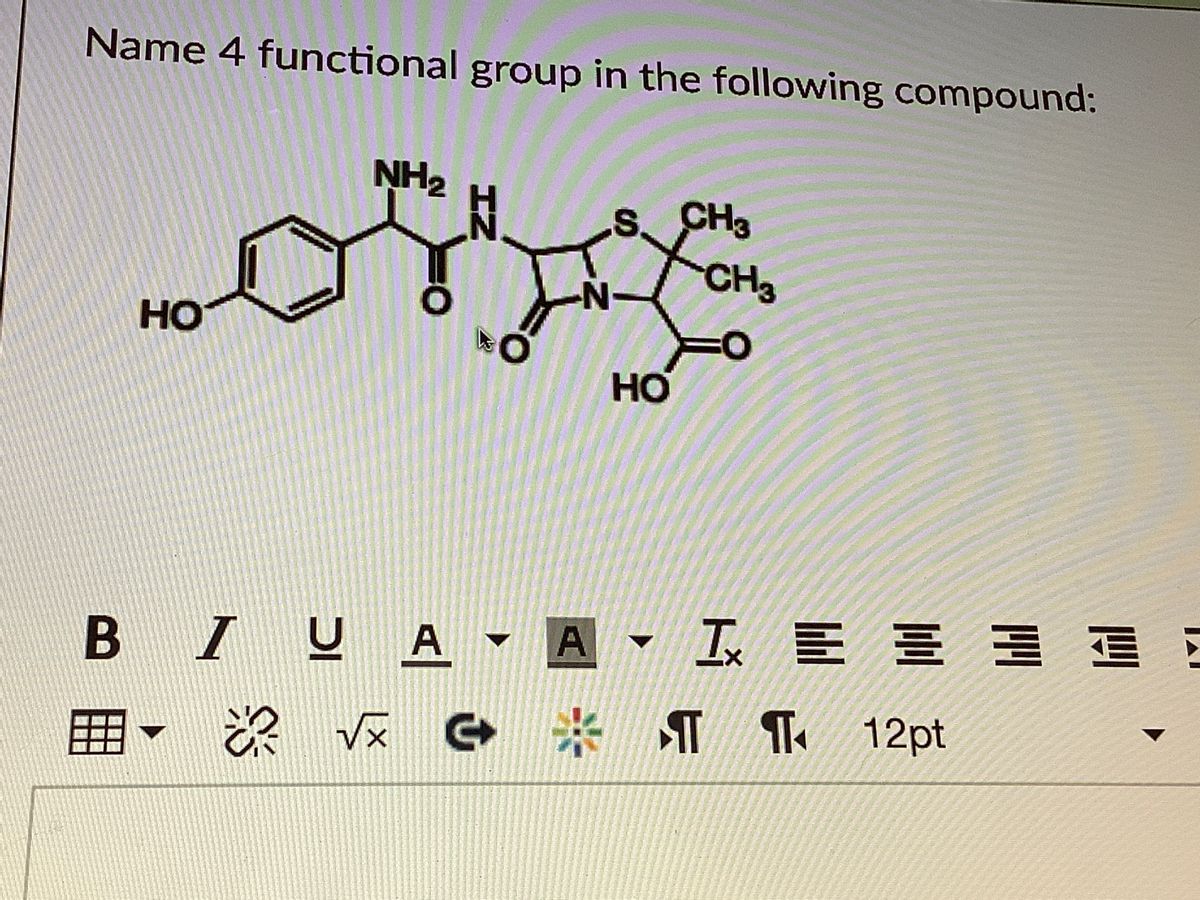
functionalization: Addition of specific functional groups to afford the compound new, desirable properties.functional group: A specific grouping of elements that is characteristic of a class of compounds, and determines some properties and reactions of that class.Functional groups can be used to distinguish similar compounds from each other.Functional groups will undergo the same type of reactions regardless of the compound of which they are a part however, the presence of certain functional groups within close proximity can limit reactivity.Functional groups are often used to “functionalize” a compound, affording it different physical and chemical properties than it would have in its original form.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
